We support the creation of genome-edited strains based on our extensive experience and track record.
Since its establishment in 2018, we have completed more than 150 projects with our expertise in knock-in technology, generation of iPS cell pathological model cells by SNP conversion, and marker labeling for screening.
With our background and extensive pharmaceutical industry experience, we can provide flexible genome editing plans and services to meet your needs.
Our Track Record
POINT-1
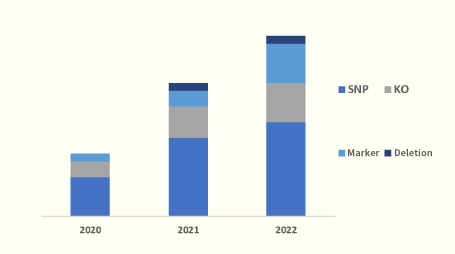
The number of genome editing projects commissioned to us has exceeded 150. The chart is a breakdown of the number of requests we have received.
The number of contracts is increasing yearly due to our ability to respond to the increasing demand and diversifying needs.
POINT-2
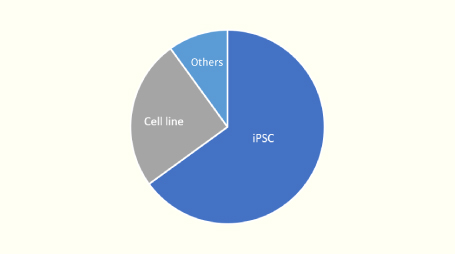
The following are the results of genome-edited cells that our company has been requested.
The majority of the genome editing requests we receive are for iPS cells.
We have been using iPS cells for genome editing since our days at Takeda, so we have abundant experience handling iPS cells.
Single Nucleotide Polymorphism (SNP) cell generation
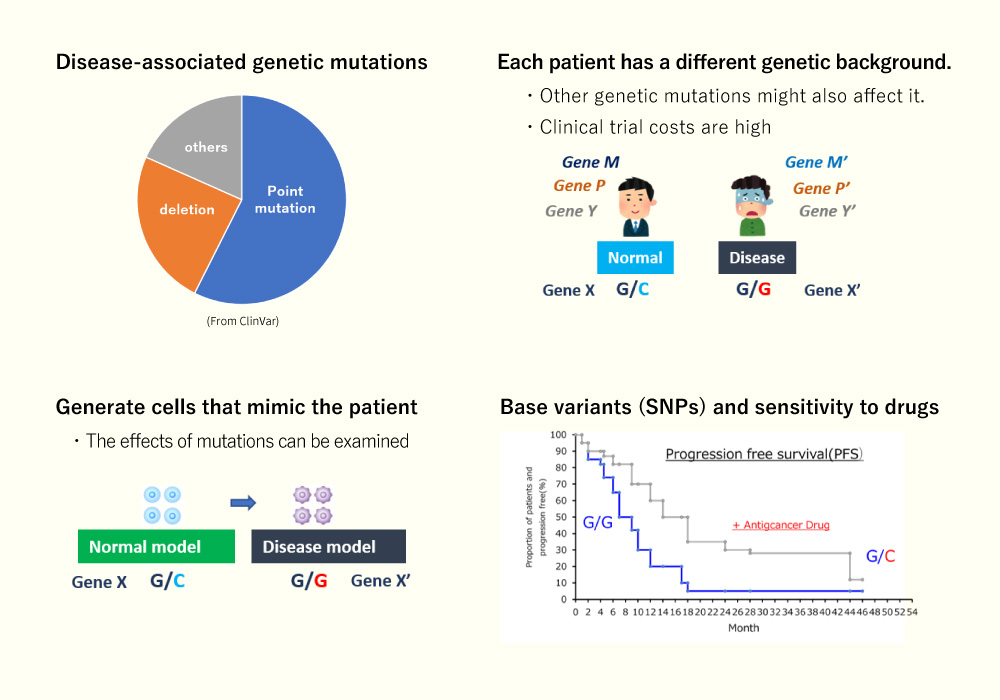
- In recent years, the relationship between diseases and genetic mutations has been well investigated through advances in genomic DNA analysis technology, and single nucleotide substitutions are involved in 60% of these cases.
- On the other hand, obtaining cell samples for each disease can be burdensome and costly for patients.
- It is known that the effects of drugs may differ depending on single nucleotide variations (SNPs) even if they do not indicate a pathological condition, and the creation of pathological model cells by genome editing using iPS cells that show pluripotency to differentiate is an indispensable tool in the early stages of drug discovery research.
Examples of single nucleotide polymorphism (SNP) cells we have produced
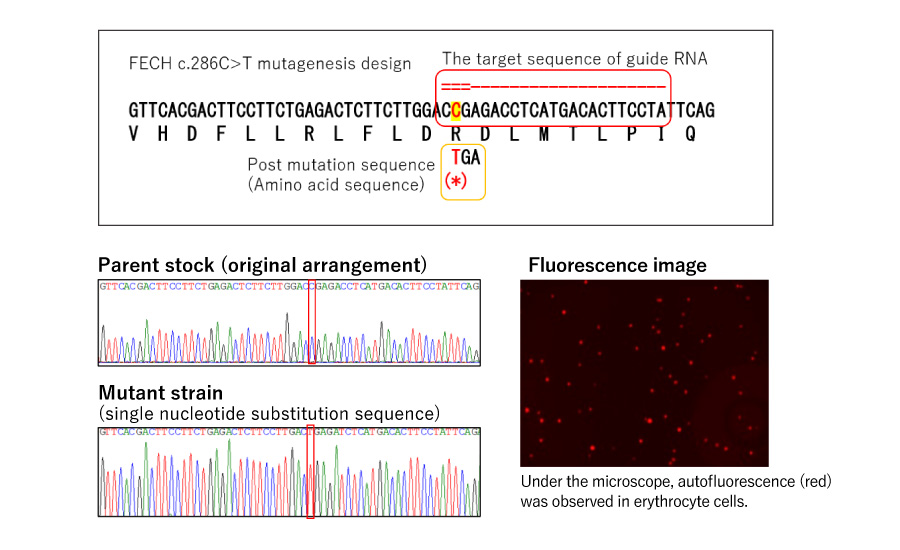
Generation of Erythropoietic protoporphyria (EPP) model cells
By introducing a point mutation (FECH c.286C>T) in one of the enzymes of the heme biosynthetic system: ferro chelatase, we confirmed autofluorescence (one of the diagnostic criteria of EPP) in erythrocytes.
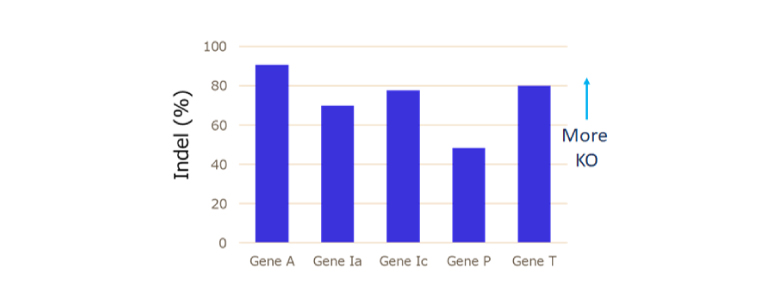
Knockout (KO) cell generation
- Conceptual (PoC) validation using a gene’s functional deletion (KO) strain is used when you wish to analyze the function of a gene.
- Due to its high efficiency, CRISPR/Cas9 can efficiently KO multiple genes at once by carefully selecting guide RNAs.
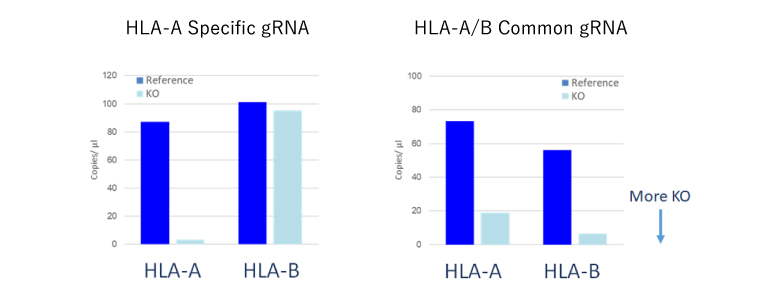
- It is also possible to KO family proteins with similar sequences simultaneously or individually.
Examples of knockout cell production we have done
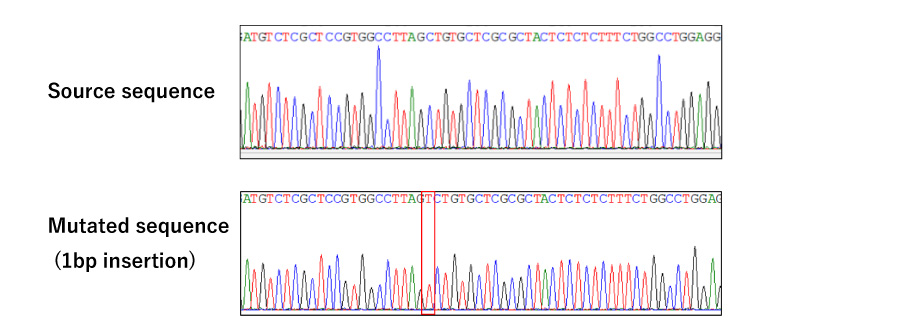
Simple KO:Efficient generation of mutagenic strains for target genes
Multiple KO:Save Time and Passage Number
Family Protein KO
Inducible KO
Floxed mice are used to make KO mice for lethal genes. iPS cells are suitable for genome editing, but once differentiated, they become reluctant in transfection.
Therefore the construction of Floxed iPS cells before differentiation can be a solution to explore the gene function in any differentiated cell type.

Knock-in KO
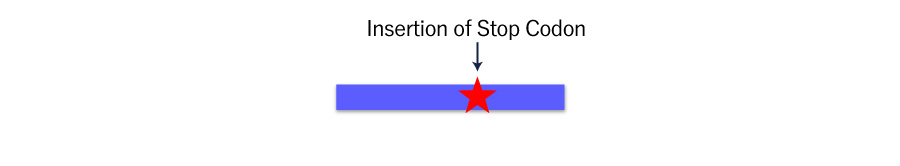
When most of the pooled cells have been disrupted the target sequence, we can explore the KO phenotype, assuming most of the disruption as KO, although KO clones can also be easily obtained.
However, the pattern of disruption are so diverse that it may be risky to hypothesize all mutated sequences to be KO just judged from their sequence information.
In such cases inserting a stop codon using the knock-in technique can be a solution to resolve the heterogeneity.
In addition, KO strains can be created using the same sequence when testing with other cell lines.
Knock-in cell creation (insert any sequence)
- Reporter iPS Cells for Differentiation Research
Differentiation reporter cells, generated by fusing fluorescent genes to differentiation markers, are useful for differentiation research.
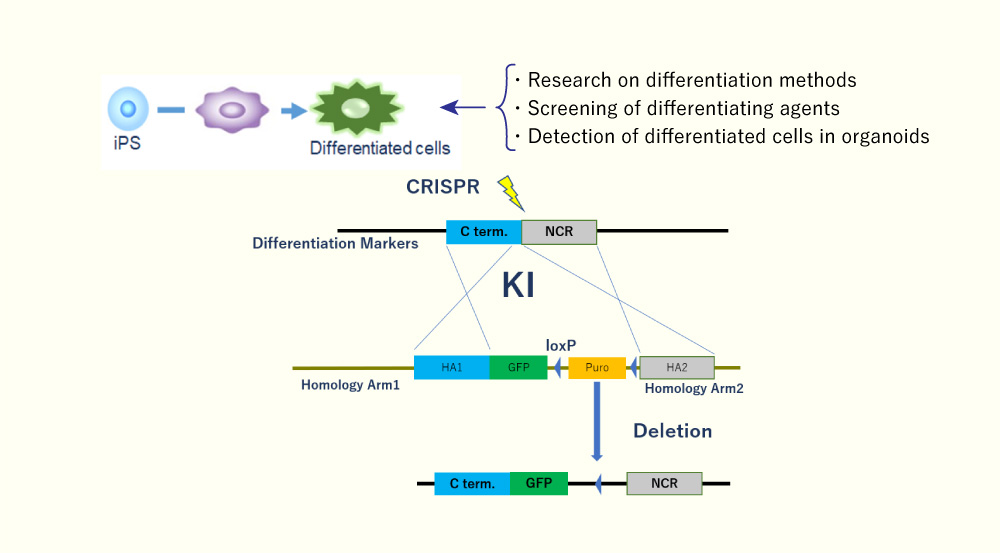
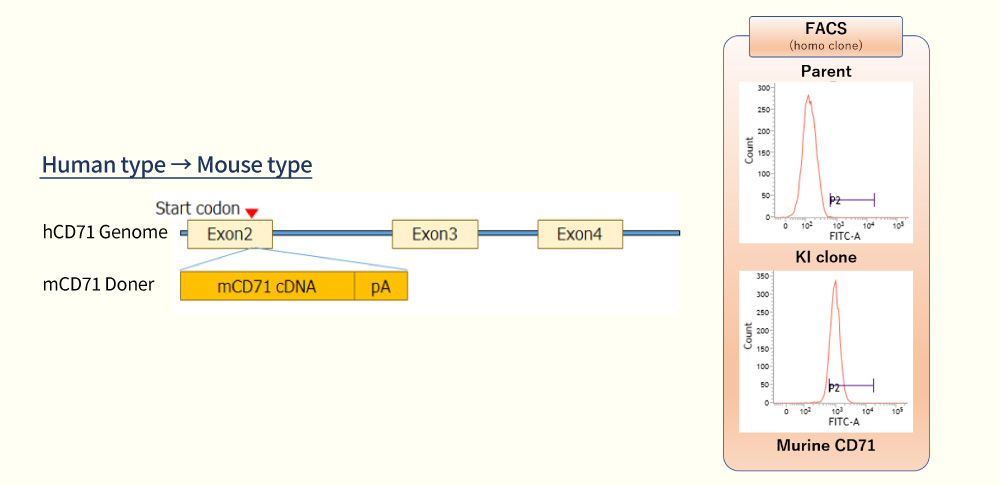
- Gene Species Conversion
Whether developing compounds are tolerated in the higher species or not is a common issue in drug development. Replacement of the target protein with another species by genome editing gives us an important information about species differences using the same cell line.
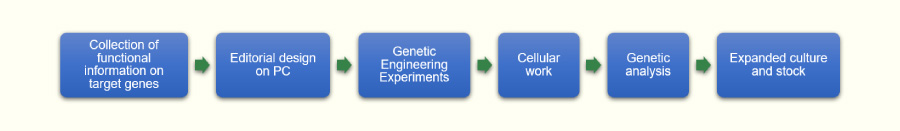
Features of GenAhead Bio
Genome-edited cells are produced through a multi-step process, the work of which needs to be tailored according to the purpose.
- Successful genome editing design
We are an experienced team with over 150 projects under our belts, which range from knockouts, marker labeling, and SNP conversion. Design is where experience counts. - Purpose-driven design
We look at the structure and function of the gene in question. We will determine which parts of the genome to target to create genome-edited cells that meet the objectives of your experiment. - Ability to tailor genome editing conditions
Clients who cannot obtain genome-edited cells often consult us despite having confidently designed guide RNAs. We have guide RNAs and donor nucleic acids that we have experience knocking in with very high efficiency, and this positive control tool sets the transfection conditions for successful genome editing as usual. - Early determination of success or failure
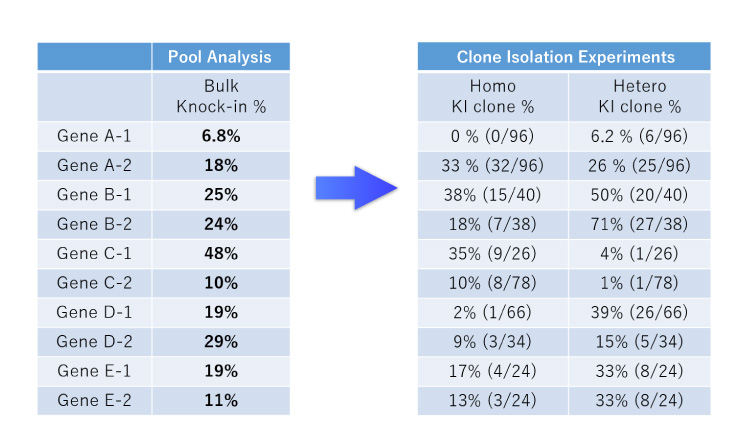
We use the CRISPR-SNIPER and Pool Analysis method (a highly sensitive and selective quantification of the final genome editing percentage in the early stages) to confirm and quantify genome editing when genome editing efficiency is very low. This makes it possible to foresee the success or failure of the experiment at an early stage.
Pool Analysis
- The success or failure of genome editing was first determined by clonal analysis, which took several months.
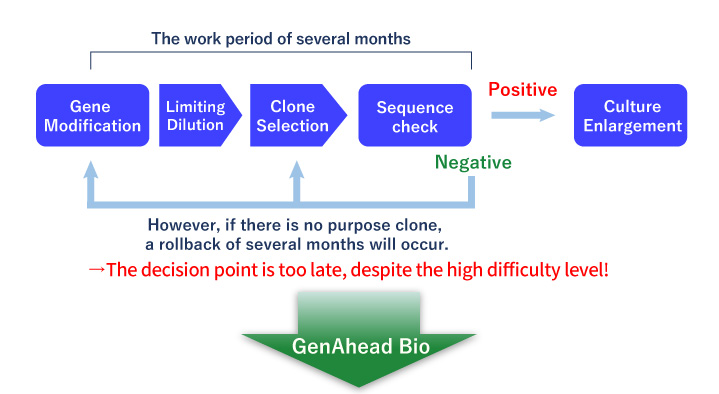
- We can quantify the genome editing efficiency by pooled analysis (CRISPR-SNIPER) to determine the probability of acquisition at an early stage.
- If the genome editing efficiency is low, we can use the vast amount of analysis data and know-how we have accumulated to optimize the conditions.
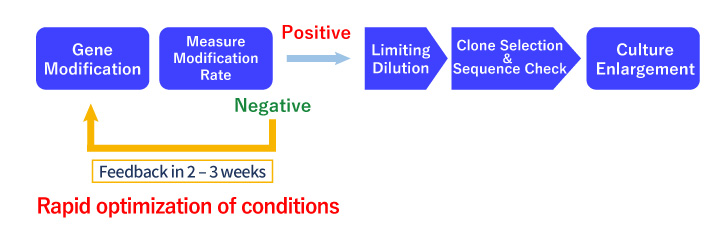
- Based on our track record of more than 150 clones isolated to date, we can offer a highly accurate Go/No Go decision based on the genome editing efficiency of the bulk cell population when proceeding to single cloning.
Licenses
- We have license agreements with ERS Genomics Ltd and BROAD INSTITUTE for research use.
- If you choose to outsource your genome editing services to us, please read the Use and Limited License.
- The Use and Limited License describes the terms and conditions for the use of the patents of both ERS Genomics Ltd and BROAD INSTITUTE, and it is understood that you have agreed to these terms and conditions when you entrust us with your genome editing work.
Collaboration with Reprocell, Inc.
We have established a cooperative system with ReproCELL Corporation, a leading company in the iPS cell field. We provide genome-edited cells using iPS cells procured by ReproCELL to many customers who do not have iPS cells or who wish to establish iPS cells.
For more information, please refer to “iPS Cell Genome Editing Service (CRISPR/Cas9)” of ReproCELL Corporation(https://reprocell.co.jp/sniper-190416)
