Our Nucleic Acid Delivery Technology
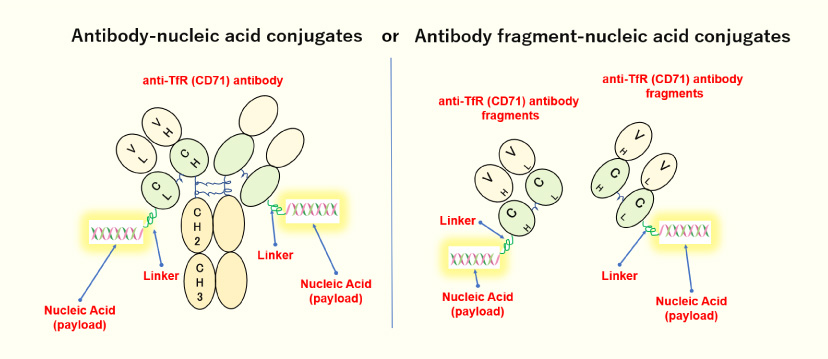
- Our proprietary technology is a conjugate which comprises an antibody or its fragment, a linker, and nucleic acids.
- By linking nucleic acids to specific antibodies or antibody fragments that recruit the nucleic acids into the target cells to harness their potential for disease treatment with their knockdown effects on causal gene expression.
- When the antibodies target a receptor of iron carrier protein; transferrin, called Transferrin Receptor (TfR = CD71), the conjugate can deliver nucleic acids to many tissues/cells, especially muscle, heart, and white blood cells,
- Our conjugates provide novel delivery method for entry into the tissues that current nucleic acid delivery methods cannot access.
About current nucleic acid delivery methods
- Adeno-Associated Virus Vector (AAV)
Nucleic acid delivery method using Adeno-Associated Virus as a vector. It cannot be used in patients with neutralizing antibodies due to immunogenicity and hepatotoxicity issues. It is estimated that 30% to 40% of the population has neutralizing antibodies. - Lipid Nanoparticle (LNP)
It is a delivery system that encapsulates nucleic acids in a non-viral lipid. Still, it is challenging to deliver to cells outside the liver by intravenous injection because it can only leak out through the sinusoidal endothelial cell pores of the liver, which are 50 nm to 200 nm in diameter. - N-Acetyl-Galactosamine (GalNAc)
A complex that binds nucleic acids to GalNAc, which shows high affinity and specificity for asialoglycoprotein receptors expressed in the liver, for uptake of nucleic acids into hepatocytes. However, it can be delivered only to the liver.
Structure of Our Antibody-Nucleic Acid Conjugate
- We were the first in the world to develop oligonucleotides delivery technology using antibodies against TfR (=CD71) (anti-TfR antibodies).
Development of Antibody-siRNA Conjugate Targeted to Cardiac and Skeletal Muscles
Journal of Controlled Release 237, p1-13, 2016.
Click Here For More Details
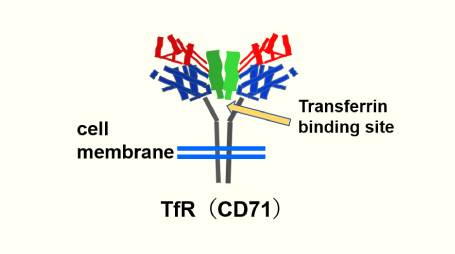
Affinity and Activity to TfR
- We have obtained a variety of anti-human TfR (CD71) antibodies that do not compete with transferrin binding. Antibodies that bind to monomers do not necessarily mean that they also bind preferentially to dimers. GenAhead antibodies recognize TfR (CD71) dimer found on the cell surface.
- The binding affinities of several anti-TfR Ab clones to epitopes A,B,B’, C, and D are shown in the table. Silencing activities were evaluated among various anti-TfR clone-conjugated anti-HPRT siRNA measuring the expression of HPRT in human cells.
The conjugates prepared from high-affinity antibodies are highlighted in the red box, suggesting that high-affinity antibodies do not always show potent silencing.
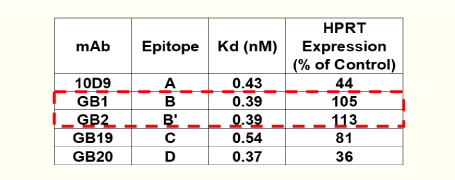
hypoxanthine phosphoribosyltransferase 1:HPRT
HPRT expression works as a good reporter for the siRNA function.
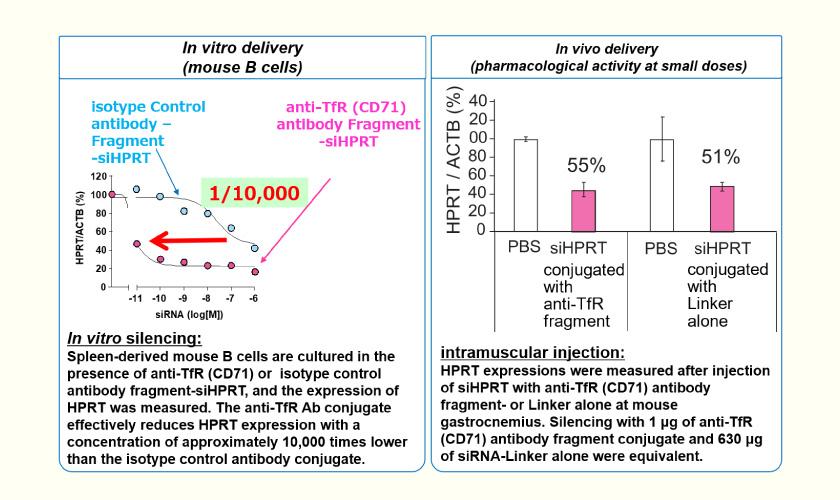
Nucleic Acid Delivery Potency
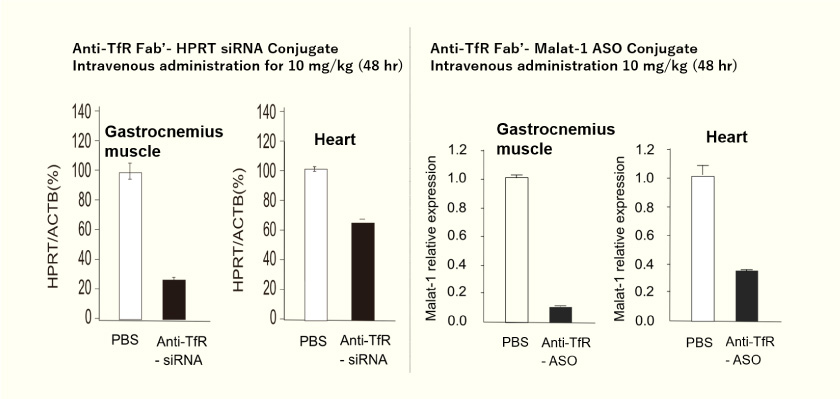
- In vitro and in vivo studies of siRNA (siHPRT) conjugate show that nucleic acids reach the cytoplasm, and that siRNA has a silencing effect.
Durability of Action
- A single intravenous dose of anti-TfR (CD71) antibody fragment – siHPRT (10 mg/kg) caused sustained silencing in the gastrocnemius and heart for one month. (siNC = negative control siRNA)
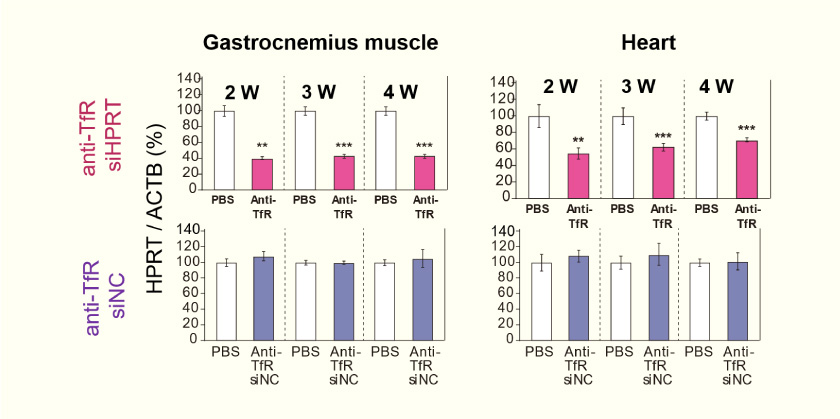
Effect of Repetition on Knockdown
- The siRNA final concentration is the same for the same number of treatments to human cell lines. The gene suppression is stronger in every third day, suggesting that the long interval gives the cells more opportunity for receptor-mediated endocytosis. Anti-human TfR antibody-siRNA shows more potent silencing in lower concentrations but less potent in higher concentrations when compared with the fragment.
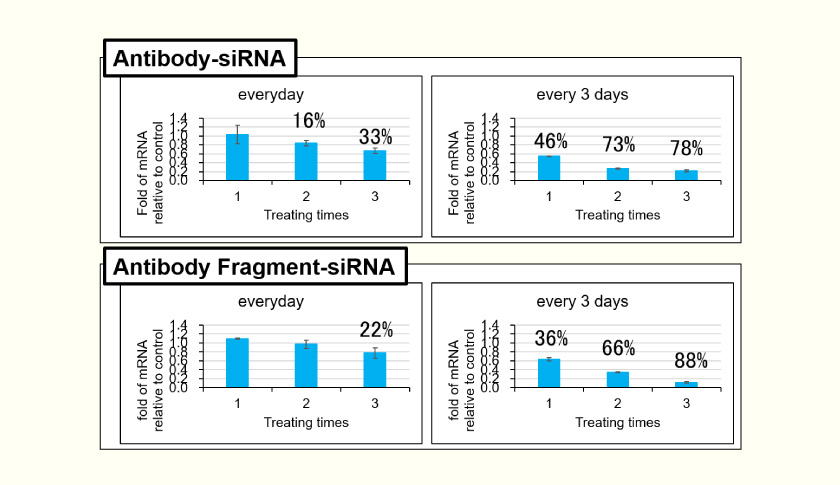
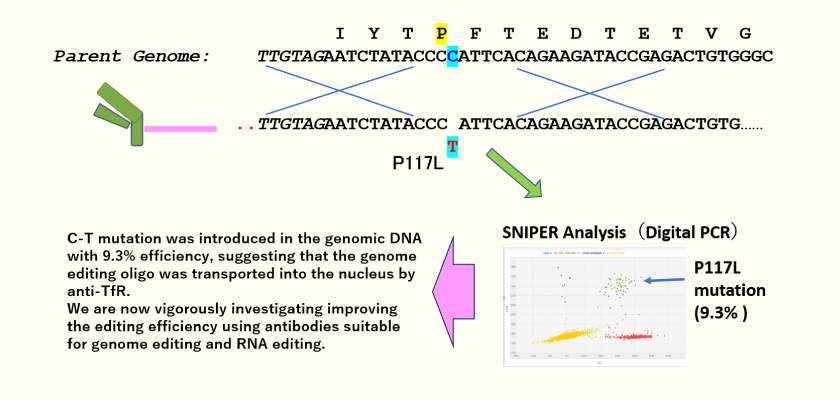
Nucleic Acid Delivery to Cell Nuclei
- Presenilin-1 (P117L) is a known mutation found in familial Alzheimer’s disease. Here we show an example of generating a cellular model of Alzheimer’s disease by introducing Presenilin-1 (P117L) donor oligonucleotide bound to an anti-TfR antibody fragment into the cells.
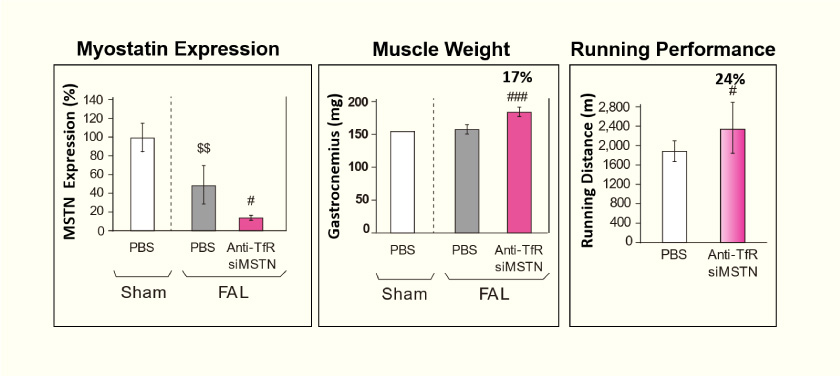
Therapeutic Efficacy in a Mouse Model of PAD Disease
- Peripheral arterial disease (PAD) occurs when blood flow in lower extremity blood vessels decreases due to aging, obesity, atherosclerosis, diabetes, and other factors. Skeletal muscles are affected, resulting in intermittent claudication that causes pain when walking and, in more advanced cases, pain at rest and even amputation of the lower limb.
- We created an animal model of a skeletal muscle disorder that recapitulates PAD by performing femoral artery ligation (FAL) in mice. Forty μg of anti-TfR antibody fragment-siRNA (target gene: myostatin) was injected weekly into the gastrocnemius muscle, and the running distances were measured four weeks later.
Nucleic Acid (Payload) Variations
- The nucleic acids to be conjugated are not only siRNAs that function in the cytoplasm, but also antisense oligonucleotides (ASO) or genome-editing oligonucleotides that function in the nucleus.
Nucleic Acid Delivery Business Development
- Licensing of anti-TfR antibody (fragment) nucleic acid conjugates
- Developmental research using anti-TfR antibody (fragment) nucleic acid conjugates
- Providing our humanized anti-hTfR antibodies
- Collaboration (partners wanted)
- Payload: mRNA, RNA/genome editing oligo, PMO, ASO, siRNA
(PMO = Phosphorodiamidate morpholino oligomers ) - Our technology
- Nucleic acid field
- siRNA design
- nucleic acid quantification of LNP samples
- internalization rate measurement
- conjugation of linkers to nucleic acids
- Antibody field
- antibody fragmentation
- antibody modification of LNP samples
- sequencing and humanization of mouse & rat monoclonal antibody variable regions
- binding of nucleic acids to antibodies provided by partners
- Nucleic acid field
- Payload: mRNA, RNA/genome editing oligo, PMO, ASO, siRNA
New Development of Antibody-Nucleic Acid Conjugates
- Expansion of target organs
- Application to viral infections
- Expansion to genome editing
